| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-07-21 20:26:17 UTC |
|---|
| Update Date | 2014-12-24 20:25:50 UTC |
|---|
| Accession Number | T3D2712 |
|---|
| Identification |
|---|
| Common Name | Caffeine |
|---|
| Class | Small Molecule |
|---|
| Description | Caffeine is the most widely consumed psychostimulant drug in the world that mostly is consumed in the form of coffee. Whether caffeine and/or coffee consumption contribute to the development of cardiovascular disease (CVD), the single leading cause of death in the US, is unclear. The literature indicates a strong relationship between boiled, unfiltered coffee consumption and elevated cholesterol levels; however, there is a critical gap in the literature regarding the effects of coffee or caffeine consumption on fibrinogen or CRP, which is an independent predictor of CVD risk. Available studies are limited by small samples sizes, inclusion of only men (or few women) and unrepresented age or ethnic groups. There is a critical need for controlled laboratory and epidemiological studies that include fibrinogen and CRP markers of CVD risk before conclusions can be drawn regarding the health effects of caffeine and/or coffee in a normal, healthy population of men and women. (19). The relationship between caffeine consumption and various illnesses such as cardiovascular disease and cancer remains equivocal. Prudence might dictate that pregnant women and chronically ill individuals exercise restraint in their use of caffeine, although research suggests relatively low or nonexistent levels of risk associated with moderate caffeine consumption. (7). There is extensive evidence that caffeine at dietary doses increases blood pressure (BP). However, concern that the drug may contribute to cardiovascular disease appears to have been dampened by (1) the belief that habitual use leads to the development of tolerance, and (2) confusion regarding relevant epidemiologic findings. When considered comprehensively, findings from experimental and epidemiologic studies converge to show that BP remains reactive to the pressor effects of caffeine in the diet. Overall, the impact of dietary caffeine on population BP levels is likely to be modest, probably in the region of 4/2 mm Hg. At these levels, however, population studies of BP indicate that caffeine use could account for premature deaths in the region of 14% for coronary heart disease and 20% for stroke. (8). Caffeine is a purine alkaloid that occurs naturally in coffee beans. At intake levels associated with coffee consumption, caffeine appears to exert most of its biological effects through the antagonism of the A1 and A2A subtypes of the adenosine receptor. Adenosine is an endogenous neuromodulator with mostly inhibitory effects, and adenosine antagonism by caffeine results in effects that are generally stimulatory. Some physiological effects associated with caffeine administration include central nervous system stimulation, acute elevation of blood pressure, increased metabolic rate, and diuresis. Caffeine concentrations in coffee beverages can be quite variable. A standard cup of coffee is often assumed to provide 100 mg of caffeine, but a recent analysis of 14 different specialty coffees purchased at coffee shops in the US found that the amount of caffeine in 8 oz (=240 ml) of brewed coffee ranged from 72 to 130 mg.Caffeine in espresso coffees ranged from 58 to 76 mg in a single shot. (9). Caffeine is a member of the methylxanthine family of drugs, and is the most widely consumed behaviourally active substance in the western world. A number of in vitro and in vivo studies have demonstrated that caffeine modulates both innate and adaptive immune responses. For instance studies indicate that caffeine and its major metabolite paraxanthine suppress neutrophil and monocyte chemotaxis, and also suppress production of the pro-inflammatory cytokine tumor necrosis factor (TNF) alpha from human blood. Caffeine has also been reported to suppress human lymphocyte function as indicated by reduced T-cell proliferation and impaired production of Th1 (interleukin [IL]-2 and interferon [IFN]-gamma), Th2 (IL-4, IL-5) and Th3 (IL-10) cytokines. Studies also indicate that caffeine suppresses antibody production. The evidence suggests that at least some of the immunomodulatory actions of caffeine are mediated via inhibition of cyclic adenosine monophosphate (cAMP)-phosphodiesterase (PDE), and consequential increase in intracellular cAMP concentrations. Overall, these studies indicate that caffeine, like other members of the methylxanthine family, is largely anti-inflammatory in nature, and based on the pharmacokinetics of caffeine, many of its immunomodulatory effects occur at concentrations that are relevant to normal human consumption. (10). |
|---|
| Compound Type | - Amide
- Anorexigenic Agent
- Appetite Depressant
- Central Nervous System Stimulant
- Drug
- Food Toxin
- Household Toxin
- Metabolite
- Natural Compound
- Organic Compound
- Phosphodiesterase Inhibitor
- Purinergic P1 Receptor Antagonist
|
|---|
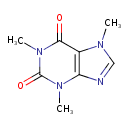
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | 1,3,7-Trimethyl-2,6-dioxopurine | | 1,3,7-Trimethyl-3,7-dihydro-1H-purine-2,6-dione | | 1,3,7-trimethylpurine-2,6-dione | | 1,3,7-Trimethylxanthine | | 1-Methyl-Theobromine | | 1-methyltheobromine | | 3,7-Dihydro-1,3,7-trimethyl-1H-purin-2,6-dion | | 3,7-Dihydro-1,3,7-trimethyl-1H-purine-2,6-dione | | 7-Methyl Theophylline | | 7-methyltheophylline | | Anhydrous caffeine | | Anhydrous caffeine (JP15) | | Cafcit | | Cafeína | | Caféine | | Caffedrine | | Coffein | | Dexitac | | Durvitan | | Enerjets | | Guaranine | | Hycomine | | Koffein | | Lanorinal | | Mateína | | Methyltheobromide | | Methyltheobromine | | Methylxanthine theophylline | | Monohydrate Caffeine | | No-Doz | | Pep-Back | | Quick Pep | | Teina | | Thein | | Theine | | Vivarin | | Wake-Up |
|
|---|
| Chemical Formula | C8H10N4O2 |
|---|
| Average Molecular Mass | 194.191 g/mol |
|---|
| Monoisotopic Mass | 194.080 g/mol |
|---|
| CAS Registry Number | 58-08-2 |
|---|
| IUPAC Name | 1,3,7-trimethyl-2,3,6,7-tetrahydro-1H-purine-2,6-dione |
|---|
| Traditional Name | caffeine |
|---|
| SMILES | CN1C=NC2=C1C(=O)N(C)C(=O)N2C |
|---|
| InChI Identifier | InChI=1S/C8H10N4O2/c1-10-4-9-6-5(10)7(13)12(3)8(14)11(6)2/h4H,1-3H3 |
|---|
| InChI Key | InChIKey=RYYVLZVUVIJVGH-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as xanthines. These are purine derivatives with a ketone group conjugated at carbons 2 and 6 of the purine moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Imidazopyrimidines |
|---|
| Sub Class | Purines and purine derivatives |
|---|
| Direct Parent | Xanthines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Xanthine
- Purinone
- 6-oxopurine
- Alkaloid or derivatives
- Pyrimidone
- Pyrimidine
- N-substituted imidazole
- Heteroaromatic compound
- Vinylogous amide
- Imidazole
- Azole
- Urea
- Lactam
- Azacycle
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | - Kidney
- Liver
- Prostate
- Skin
|
|---|
| Pathways | |
|---|
| Applications | |
|---|
| Biological Roles | |
|---|
| Chemical Roles | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | White powder. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 238°C | | Boiling Point | Not Available | | Solubility | 2.16E+004 mg/L (at 25°C) | | LogP | -0.07 |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| GC-MS | GC-MS Spectrum - GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (0 TMS) | splash10-0536-3900000000-a9e112713ffae6dabdaa | 2014-06-16 | View Spectrum | | GC-MS | GC-MS Spectrum - GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (0 TMS) | splash10-0536-2900000000-8cdcd005b2e7622a02a3 | 2014-06-16 | View Spectrum | | GC-MS | GC-MS Spectrum - GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (Non-derivatized) | splash10-052f-0900000000-f1084acfddb240696073 | 2014-06-16 | View Spectrum | | GC-MS | GC-MS Spectrum - GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (Non-derivatized) | splash10-05nf-6900000000-8670a644cee5d9de78d4 | 2014-06-16 | View Spectrum | | GC-MS | GC-MS Spectrum - GC-MS (Non-derivatized) | splash10-0536-3900000000-4430852b279a72e34822 | 2014-06-16 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0006-0900000000-51898e93480e848d7da1 | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - CI-B (Non-derivatized) | splash10-0002-0900000000-2aed5d425b6a95add5db | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0a4l-4900000000-3ff72dace6687d242f1f | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0006-1900000000-2ba1fae6e27c7b836984 | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - CI-B (Non-derivatized) | splash10-0002-0900000000-fd859aeb416e320d6379 | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - GC-EI-TOF (Non-derivatized) | splash10-0536-3900000000-a9e112713ffae6dabdaa | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - GC-EI-TOF (Non-derivatized) | splash10-0536-2900000000-8cdcd005b2e7622a02a3 | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - GC-EI-TOF (Non-derivatized) | splash10-052f-0900000000-f1084acfddb240696073 | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - GC-EI-TOF (Non-derivatized) | splash10-05nf-6900000000-8670a644cee5d9de78d4 | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - GC-MS (Non-derivatized) | splash10-0536-3900000000-4430852b279a72e34822 | 2017-09-12 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-052r-0900000000-41f36d541d34d2088964 | 2017-07-27 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-0006-0900000000-447fc72b2c709e2e18a9 | 2012-07-24 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-000i-1900000000-5e3b29de16ad91c79fe0 | 2012-07-24 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-0006-9100000000-d6f6c52ac36c8f25a500 | 2012-07-24 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI M-80) , Positive | splash10-0006-0900000000-cddd24399d942b1ac97c | 2012-08-31 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - CI-B (Unknown) , Positive | splash10-0002-0900000000-2aed5d425b6a95add5db | 2012-08-31 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI M-60) , Positive | splash10-0a4l-4900000000-3ff72dace6687d242f1f | 2012-08-31 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI M-68) , Positive | splash10-0006-1900000000-2ba1fae6e27c7b836984 | 2012-08-31 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - CI-B (HITACHI M-60) , Positive | splash10-0002-0900000000-63b9ef42a3e8d59e9997 | 2012-08-31 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Positive | splash10-0002-0900000000-185b3d97d8857a0f269d | 2012-08-31 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Positive | splash10-0002-0900000000-f8a0c0dd9f5c4a272eaf | 2012-08-31 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Positive | splash10-000i-1900000000-dd8e35226d0704aa657d | 2012-08-31 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Positive | splash10-01x9-9800000000-70e3b0eb52481c39d191 | 2012-08-31 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Positive | splash10-001l-9100000000-6d428a5571beb0e3fed4 | 2012-08-31 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Positive | splash10-0002-0900000000-98bec16f898808c3de68 | 2012-08-31 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) 30V, Positive | splash10-0002-0900000000-b112e4e059e1ecf98c5f | 2012-08-31 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - DI-ESI-qTof , Positive | splash10-00dr-0900000000-42c6f8fc7b924e9c64f3 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-qTof , Positive | splash10-000i-4900000000-a60a480f1340558740a2 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT , positive | splash10-000i-0900000000-695d910d49fc0beb1d54 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT , positive | splash10-0002-0900000000-094879886a2e72bf0c56 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT , positive | splash10-0002-0900000000-fa38c865089a3a05f287 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT , positive | splash10-000b-0900000000-0e82732a924c974dd0c8 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT , positive | splash10-000i-0900000000-bfc94c8091471847482b | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT , positive | splash10-000i-1900000000-c8fcf16986c494898203 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT , positive | splash10-000i-3900000000-9569e0552abb7ebd145a | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT , positive | splash10-0002-0900000000-3a924abd44877050c1c9 | 2017-09-14 | View Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-052f-8900000000-68b5e9aa3404fb3d8d3a | 2014-09-20 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, CDCl3, experimental) | Not Available | 2012-12-04 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 90 MHz, CDCl3, experimental) | Not Available | 2014-09-20 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 25.16 MHz, CDCl3, experimental) | Not Available | 2014-09-23 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, CDCl3, experimental) | Not Available | 2021-10-10 | View Spectrum | | 2D NMR | [1H, 1H]-TOCSY. Unexported temporarily by An Chi on Oct 15, 2021 until json or nmrML file is generated. 2D NMR Spectrum (experimental) | Not Available | 2012-12-04 | View Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, CDCl3, experimental) | Not Available | 2012-12-05 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Readily absorbed after oral or parenteral administration. The peak plasma level for caffeine range from 6-10mg/L and the mean time to reach peak concentration ranged from 30 minutes to 2 hours. |
|---|
| Mechanism of Toxicity | Caffeine stimulates medullary, vagal, vasomotor, and respiratory centers, promoting bradycardia, vasoconstriction, and increased respiratory rate. This action was previously believed to be due primarily to increased intracellular cyclic 3′,5′-adenosine monophosphate (cyclic AMP) following inhibition of phosphodiesterase, the enzyme that degrades cyclic AMP. It is now thought that xanthines such as caffeine act as antagonists at adenosine-receptors within the plasma membrane of virtually every cell. As adenosine acts as an autocoid, inhibiting the release of neurotransmitters from presynaptic sites but augmenting the actions of norepinephrine or angiotensin, antagonism of adenosine receptors promotes neurotransmitter release. This explains the stimulatory effects of caffeine. Blockade of the adenosine A1 receptor in the heart leads to the accelerated, pronounced "pounding" of the heart upon caffeine intake. |
|---|
| Metabolism | Hepatic cytochrome P450 1A2 (CYP 1A2) is involved in caffeine biotransformation. About 80% of a dose of caffeine is metabolized to paraxanthine (1,7-dimethylxanthine), 10% to theobromine (3,7-dimethylxanthine), and 4% to theophylline (1,3-dimethylxanthine).
Route of Elimination: In young infants, the elimination of caffeine is much slower than that in adults due to immature hepatic and/or renal function.
Half Life: 3 to 7 hours in adults, 65 to 130 hours in neonates |
|---|
| Toxicity Values | LD50: 127 mg/kg (Oral, Mouse) (6) |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | 3, not classifiable as to its carcinogenicity to humans. (23) |
|---|
| Uses/Sources | Caffeine is a central nervous system (CNS) stimulant, having the effect of temporarily warding off drowsiness and restoring alertness. For management of fatigue, orthostatic hypotension, and for the short term treatment of apnea of prematurity in infants. |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Caffeine may increase rates of miscarriage and low birth weight.
Caffeine withdrawal symptoms include fatigue, headache, nausea and irritability. Using large amounts of these drugs can result in a condition known as amphetamine psychosis -- which can result in auditory, visual and tactile hallucinations, intense paranoia, irrational thoughts and beliefs, delusions, and mental confusion. |
|---|
| Symptoms | High doses may cause nausea, diarrhea, insomnia, headaches, nervousness or agitation, and the shakes. |
|---|
| Treatment | Convulsions may be treated with IV administration of diazepam or a barbiturate such as pentobarbital sodium. (22) |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB00201 |
|---|
| HMDB ID | HMDB01847 |
|---|
| PubChem Compound ID | 2519 |
|---|
| ChEMBL ID | CHEMBL113 |
|---|
| ChemSpider ID | 2424 |
|---|
| KEGG ID | C07481 |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | 27732 |
|---|
| BioCyc ID | 1-3-7-TRIMETHYLXANTHINE |
|---|
| CTD ID | Not Available |
|---|
| Stitch ID | Caffeine |
|---|
| PDB ID | CFF |
|---|
| ACToR ID | 235 |
|---|
| Wikipedia Link | Caffeine |
|---|
| References |
|---|
| Synthesis Reference | Kaspar Bott, “Preparation of caffeine.” U.S. Patent US4380631, issued December, 1976. |
|---|
| MSDS | Link |
|---|
| General References | - Nathanson JA: Caffeine and related methylxanthines: possible naturally occurring pesticides. Science. 1984 Oct 12;226(4671):184-7. [6207592 ]
- Haskell CF, Kennedy DO, Wesnes KA, Milne AL, Scholey AB: A double-blind, placebo-controlled, multi-dose evaluation of the acute behavioural effects of guarana in humans. J Psychopharmacol. 2007 Jan;21(1):65-70. Epub 2006 Mar 13. [16533867 ]
- Smit HJ, Gaffan EA, Rogers PJ: Methylxanthines are the psycho-pharmacologically active constituents of chocolate. Psychopharmacology (Berl). 2004 Nov;176(3-4):412-9. Epub 2004 May 5. [15549276 ]
- Benjamin LT Jr, Rogers AM, Rosenbaum A: Coca-Cola, caffeine, and mental deficiency: Harry Hollingworth and the Chattanooga trial of 1911. J Hist Behav Sci. 1991 Jan;27(1):42-55. [2010614 ]
- Nehlig A, Daval JL, Debry G: Caffeine and the central nervous system: mechanisms of action, biochemical, metabolic and psychostimulant effects. Brain Res Brain Res Rev. 1992 May-Aug;17(2):139-70. [1356551 ]
- Wishart DS, Knox C, Guo AC, Cheng D, Shrivastava S, Tzur D, Gautam B, Hassanali M: DrugBank: a knowledgebase for drugs, drug actions and drug targets. Nucleic Acids Res. 2008 Jan;36(Database issue):D901-6. Epub 2007 Nov 29. [18048412 ]
- Lamarine RJ: Selected health and behavioral effects related to the use of caffeine. J Community Health. 1994 Dec;19(6):449-66. [7844249 ]
- James JE: Critical review of dietary caffeine and blood pressure: a relationship that should be taken more seriously. Psychosom Med. 2004 Jan-Feb;66(1):63-71. [14747639 ]
- Higdon JV, Frei B: Coffee and health: a review of recent human research. Crit Rev Food Sci Nutr. 2006;46(2):101-23. [16507475 ]
- Horrigan LA, Kelly JP, Connor TJ: Immunomodulatory effects of caffeine: friend or foe? Pharmacol Ther. 2006 Sep;111(3):877-92. Epub 2006 Mar 15. [16540173 ]
- Miyake Y, Sakaguchi K, Iwasaki Y, Ikeda H, Makino Y, Kobashi H, Araki Y, Ando M, Kita K, Shiratori Y: New prognostic scoring model for liver transplantation in patients with non-acetaminophen-related fulminant hepatic failure. Transplantation. 2005 Oct 15;80(7):930-6. [16249741 ]
- Wilkinson SC, Maas WJ, Nielsen JB, Greaves LC, van de Sandt JJ, Williams FM: Interactions of skin thickness and physicochemical properties of test compounds in percutaneous penetration studies. Int Arch Occup Environ Health. 2006 May;79(5):405-13. Epub 2006 Jan 25. [16435152 ]
- Spiller HA, Winter ML, Klein-Schwartz W, Bangh SA: Efficacy of activated charcoal administered more than four hours after acetaminophen overdose. J Emerg Med. 2006 Jan;30(1):1-5. [16434328 ]
- Ayotte P, Dewailly E, Lambert GH, Perkins SL, Poon R, Feeley M, Larochelle C, Pereg D: Biomarker measurements in a coastal fish-eating population environmentally exposed to organochlorines. Environ Health Perspect. 2005 Oct;113(10):1318-24. [16203240 ]
- Shah S, Budev M, Blazey H, Fairbanks K, Mehta A: Hepatic veno-occlusive disease due to tacrolimus in a single-lung transplant patient. Eur Respir J. 2006 May;27(5):1066-8. [16707401 ]
- Larson AM, Polson J, Fontana RJ, Davern TJ, Lalani E, Hynan LS, Reisch JS, Schiodt FV, Ostapowicz G, Shakil AO, Lee WM: Acetaminophen-induced acute liver failure: results of a United States multicenter, prospective study. Hepatology. 2005 Dec;42(6):1364-72. [16317692 ]
- Septer S, Thompson ES, Willemsen-Dunlap A: Anesthesia concerns for children with tuberous sclerosis. AANA J. 2006 Jun;74(3):219-25. [16786916 ]
- Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM: Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009 Feb 12;457(7231):910-4. doi: 10.1038/nature07762. [19212411 ]
- Rodrigues IM, Klein LC: Boiled or filtered coffee? Effects of coffee and caffeine on cholesterol, fibrinogen and C-reactive protein. Toxicol Rev. 2006;25(1):55-69. [16856769 ]
- Drugs.com [Link]
- About.com (2009). Health Effects of Drug Use. [Link]
- RxList: The Internet Drug Index (2009). [Link]
- International Agency for Research on Cancer (2014). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. [Link]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | | Gene | Gene Symbol | Gene ID | Interaction | Chromosome | Details |
|---|
|
|---|
| Down-Regulated Genes | | Gene | Gene Symbol | Gene ID | Interaction | Chromosome | Details |
|---|
|
|---|